AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
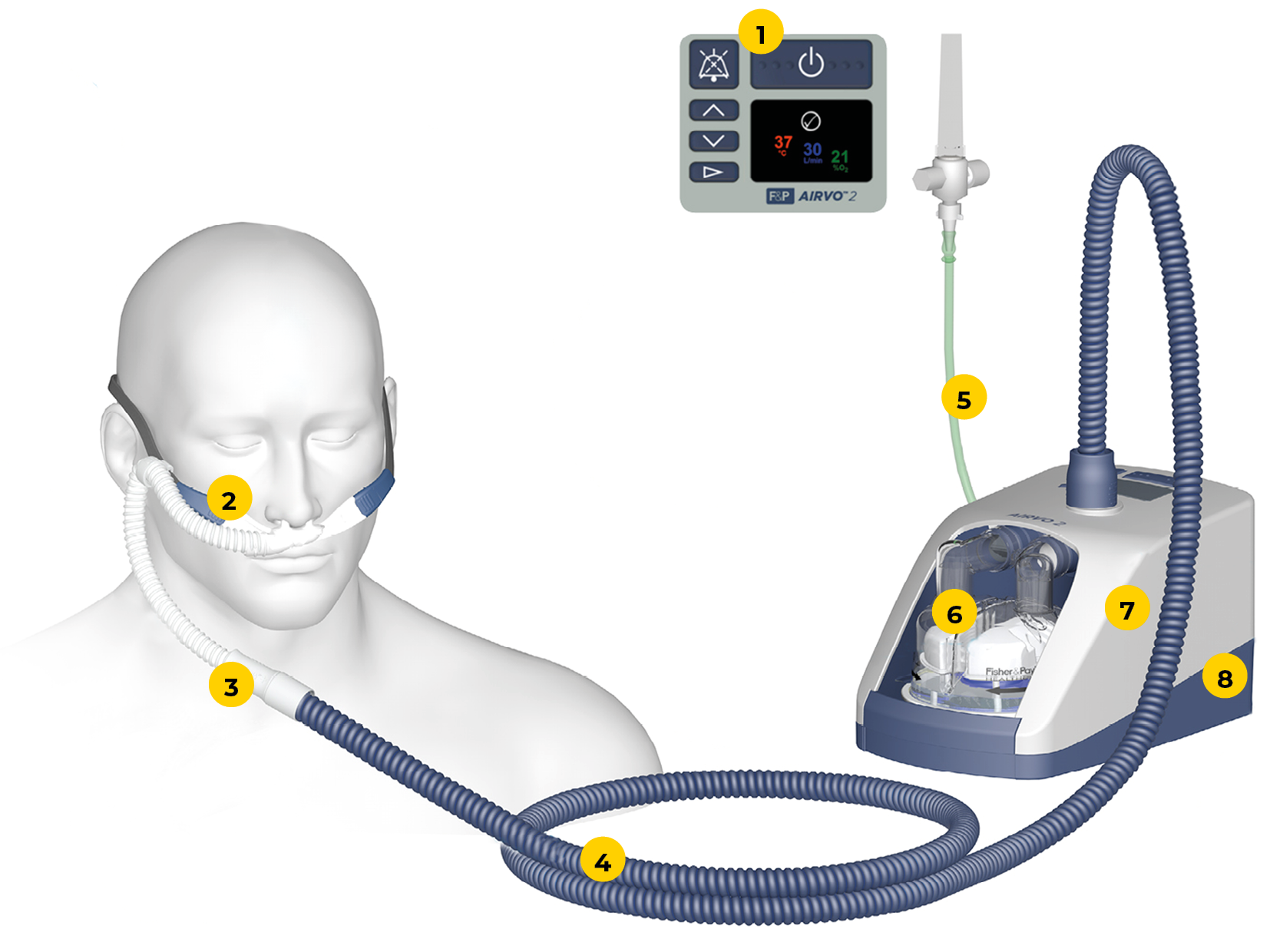
Airvo high flow8/5/2023  The indications, initiation and weaning pathways are described in the appendixes. High Flow Nasal Cannula (HFNC) therapy has been shown in studies to reduce intubation rates in infants and children with acute respiratory failure. The AIRVO 2 is a humidifier with an integrated flow generator that delivers high-flow warmed and humidified respiratory gases to spontaneously breathing. The Airvo 2 device features a humidifier with integrated flow source that delivers high flows of air/oxygen mixtures to spontaneously breathing patients. AIRVO 2 can more accurately deliver prescribed oxygen concentrations at high flows, providing both versatility and continuity of care. With AIRVO 2, the aim is to meet or exceed the patient’s normal Peak Inspiratory Demand, which creates minimal air dilution, even when breathing orally. 18)), including those who have had their upper airway bypassed. When compared with Airvo tube 900PT501 in internal F&P testing. 1-4 The myAirvo 2 is an innovative humidified high flow system that delivers the. The device can be used on infant to adult sized patients (if using on a patient greater than 10 years old, please refer to Appendix 4/5 – High Flow and the adult patient (p. Humidified high flow therapy has been shown to ease symptoms such as frequent coughs and breathlessness as well as reduce escalation of care in patients with conditions such as COPD. The AIRVO is capable of delivering supplemental oxygen at any concentration between 21% and 95%. The device may be set from 2 – 60 L/minute, depending on the interface, and delivers a flow of gas that has high levels of humidity which aids patient comfort and can enhance mucociliary clearance of secretions. The AIRVO 2 is for the treatment of spontaneously breathing patients who would benefit from receiving high flow warmed and humidified respiratory gases. The purpose of this document is to detail the process for clinical guidelines for high flow oxygen therapy on the Medical Assessment Unit and Glossop Ward. From a safety perspective no convincing adverse effects have been documented, although, similarly to nCPAP ‘abdominal distension remains a risk and requires careful monitoring. The evidence indicates that the increase in Peek Expiratory End Pressure generated is similar to airway pressure achieved by nCPAP. Much of the literature and research looking at ‘High-Flow’ therapy agree that therapy is well tolerated and with less associated nasal trauma than nasal CPAP (nCPAP). 
The high flow (up to 60 l/min) is generated by a turbine and the system. The AIRVO 2 is a humidifier with an integrated flow generator that delivers high-flow warmed and humidified respiratory gases to spontaneously breathing patients through a variety of interfaces. Download scientific diagram Airvo 2 high-flow system (Fisher & Paikel Healthcare). compared with F&P Airvo 900PT501 breathing tube in internal Fisher & Paykel Healthcare testing. This guideline is intended for all healthcare professionals caring for patients requiring High-Flow via the AIRVO 2 within the Paediatric Intensive Care Unit (Ward 1D) Royal Hospital for Children, Glasgow, and as a guide for staff caring for these patients who are being nursed in other areas. Provides a way to deliver optimally humidified respiratory gas (Optiflow High Flow therapy) direct to a tracheostomy patient. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed